The National Agency for Food and Drugs Administration and Control, NAFDAC, has raised alarm over the circulation of suspected counterfeit antibiotic drug, Augmentin 625mg tablets in the country.
The Agency is advising the public to be vigilant and watch out for the counterfeited Augmentin 625mg tablets in circulation.
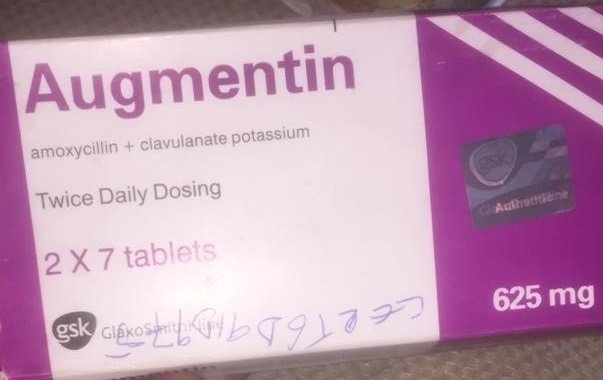
The said drug according to NAFDAC failed short of the labeling requirements.
It added that it has no inscription “manufactured by” is written on the label -only the address.

The counterfeit product manufactured date is April 2021, with batch No.: 562626 and display expiry date of April 2024 while it also claimed the medicine was approved by NAFDAC with Registration No: 04-1928.
The Mojisola Adeyeye led agency also stated that the manufacturing and expiry dates do not meet the acceptable format. It does not show No MAS scratch number for verification. The logo “gsk” is not properly positioned as on the original.
The agency insisted that the listed information indicates the product is falsified and counterfeited.
Meanwhile, NAFDAC has notified all its formations in the zones and 36 states of the federation including the FCT to carry out surveillance and mop up the falsified Augmentin tablets.
It harped on identifying the genuine Augmentin 625mg, stating that it has legible product labelling information including date markings – expiration and manufactured dates, batch number and NAFDAC registration number.
Members of the public who are in possession of the suspected counterfeit product have been advised to discontinue sale or use and submit stock to the nearest NAFDAC office, or report any suspicion of adverse drug reaction and substandard and falsified medicines on 0800-162-3322 or [email protected].


